Join us in the future of
healthcare
Vital is transforming diagnostics to ensure patients get the care they need. Provide your information and we’ll be in touch soon.



Our Data
“Above all else show the data.”
— Edward Tufte
Data speaks for itself—and that’s what makes it powerful. We seek to earn the trust of patients, providers, and the broader scientific community by showing that we take transparency as paramount. Below we share data that was collected as part of our product development process.
In addition to this data, we are actively working to publish studies on our performance, innovations, and impact in peer-reviewed journals.
Precision is in our DNA
Well, not precisely, but precision is in everything we do at Vital.
Throughout our development process, we have strived to ensure all of our studies are driven by CLSI & CLIA guidelines. Cumulatively, multiple generations of our systems have been tested across across thousands of patient samples in partnership with the world’s best lab partners.
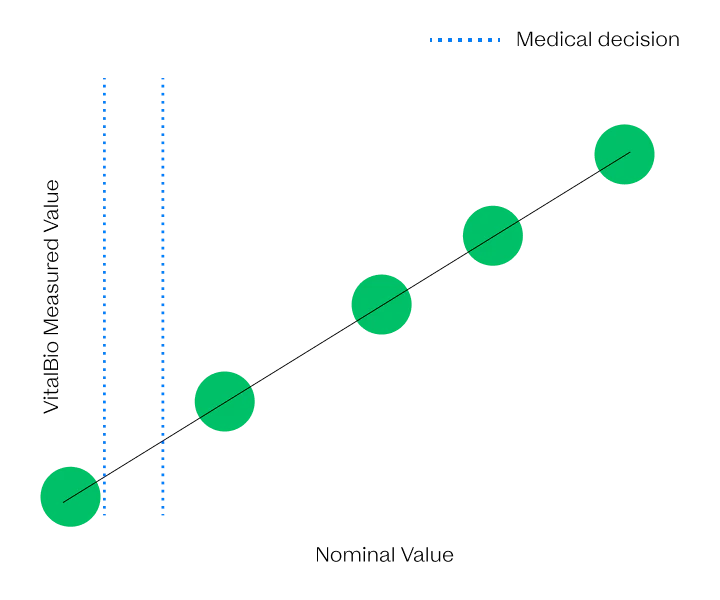
Here through the highs and lows
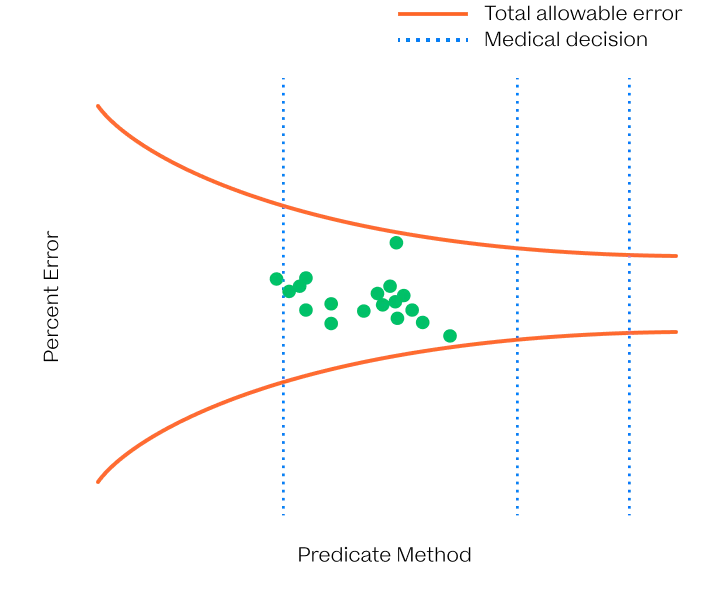
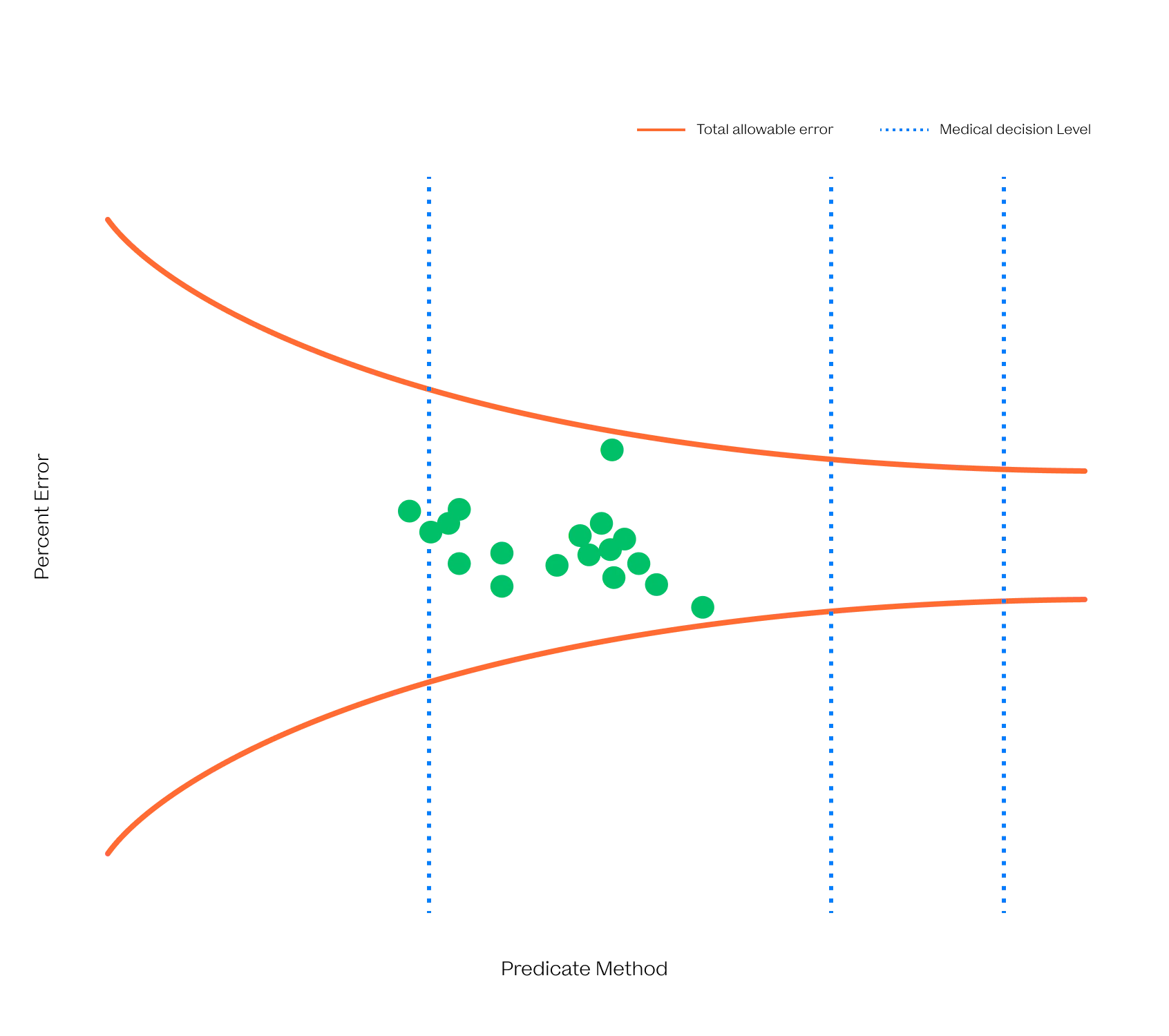
The VitalOne is intended to reliably quantify biomarkers across the physiological range.
We have developed next-generation immunoassays that are capable of ultrafast and ultrasensitive detection from a few microliters of blood with wide dynamic range. These advancements were achieved by rethinking immunoassays through the lens of biophysics.
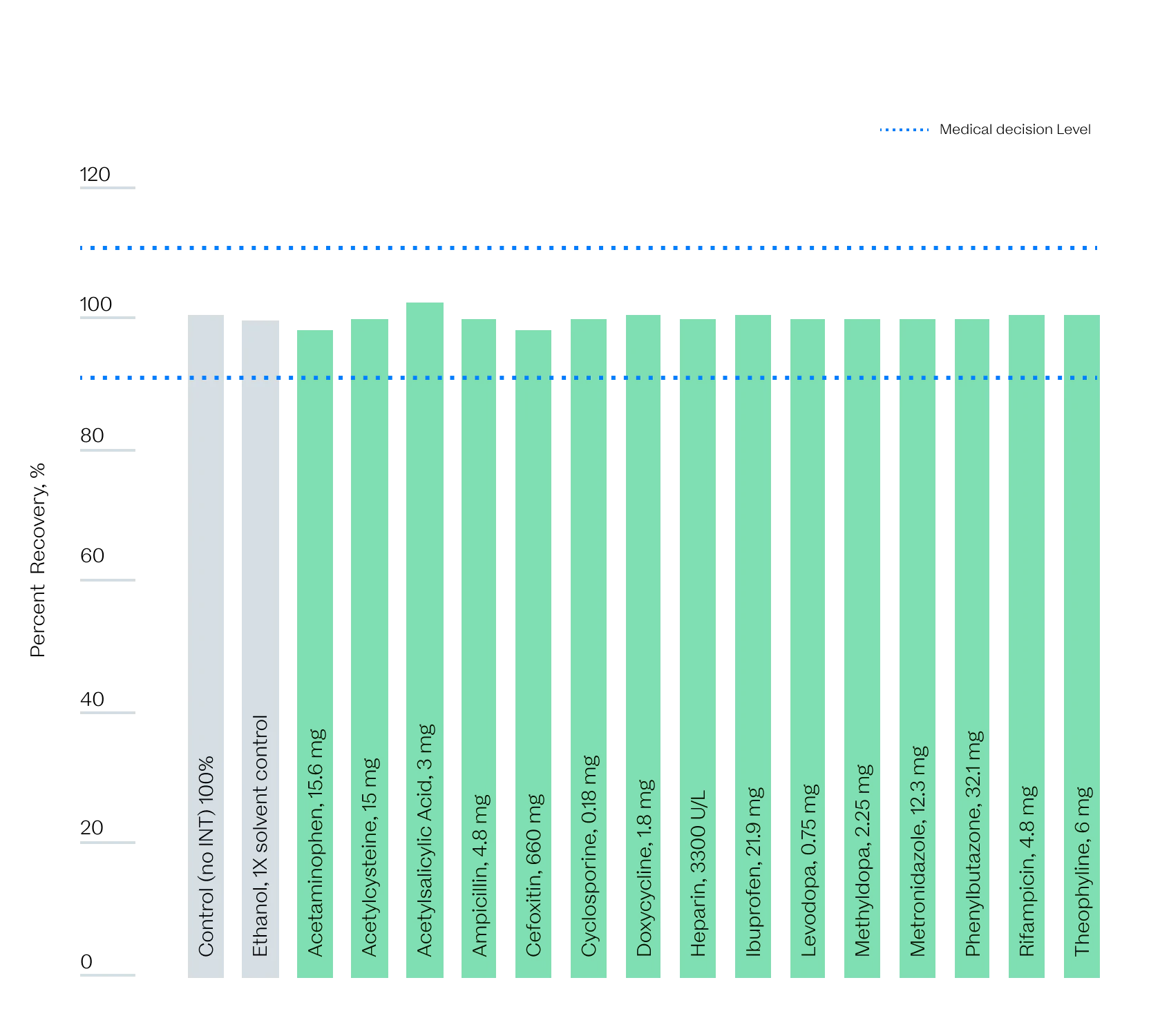
The sample is always right
We know that the results from our devices will impact patients and families—including our own.
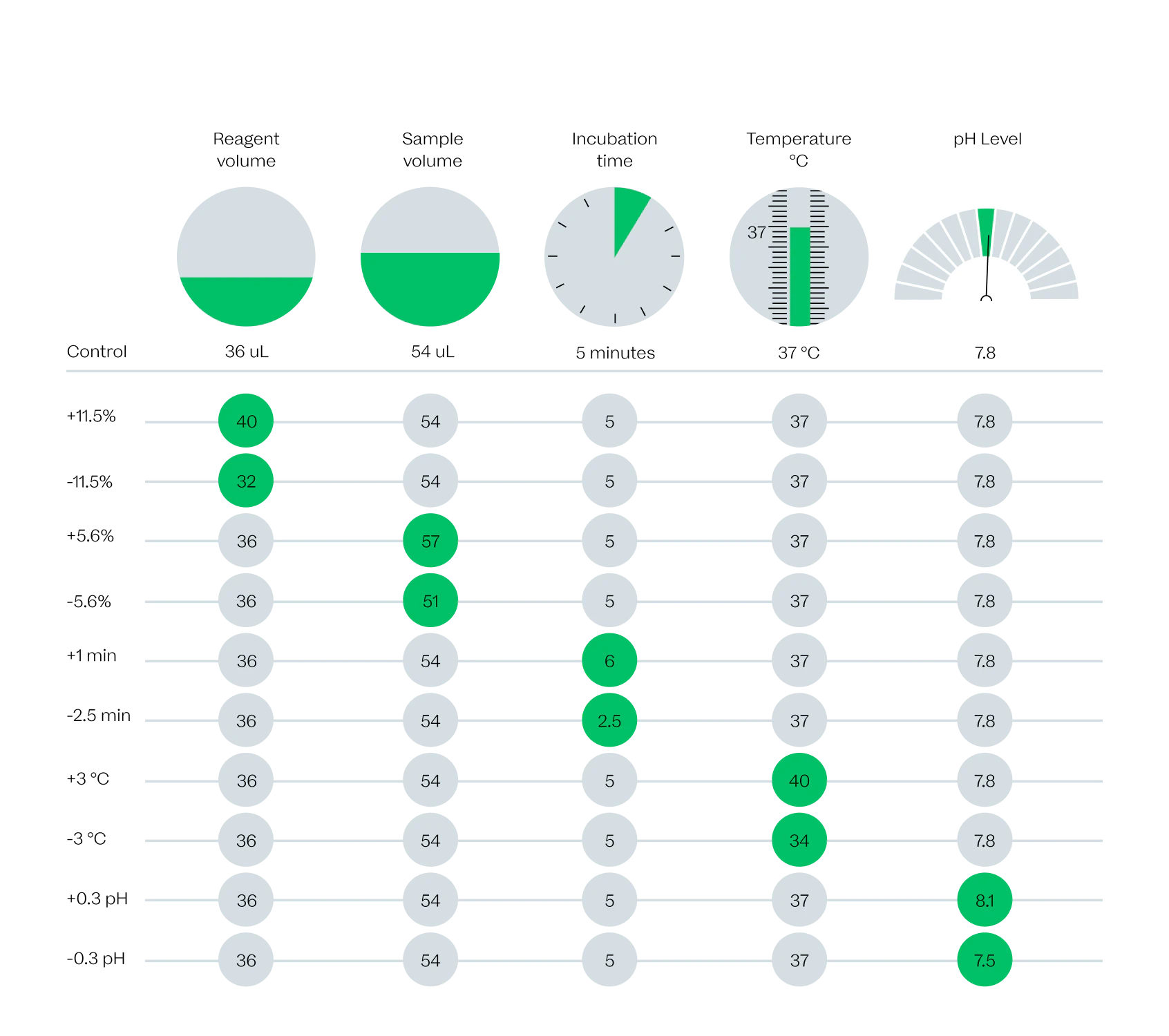
That is why we are putting every assay we are developing through a battery of tests, such as against common intereferants, to ensure that we are delivering results you can trust.

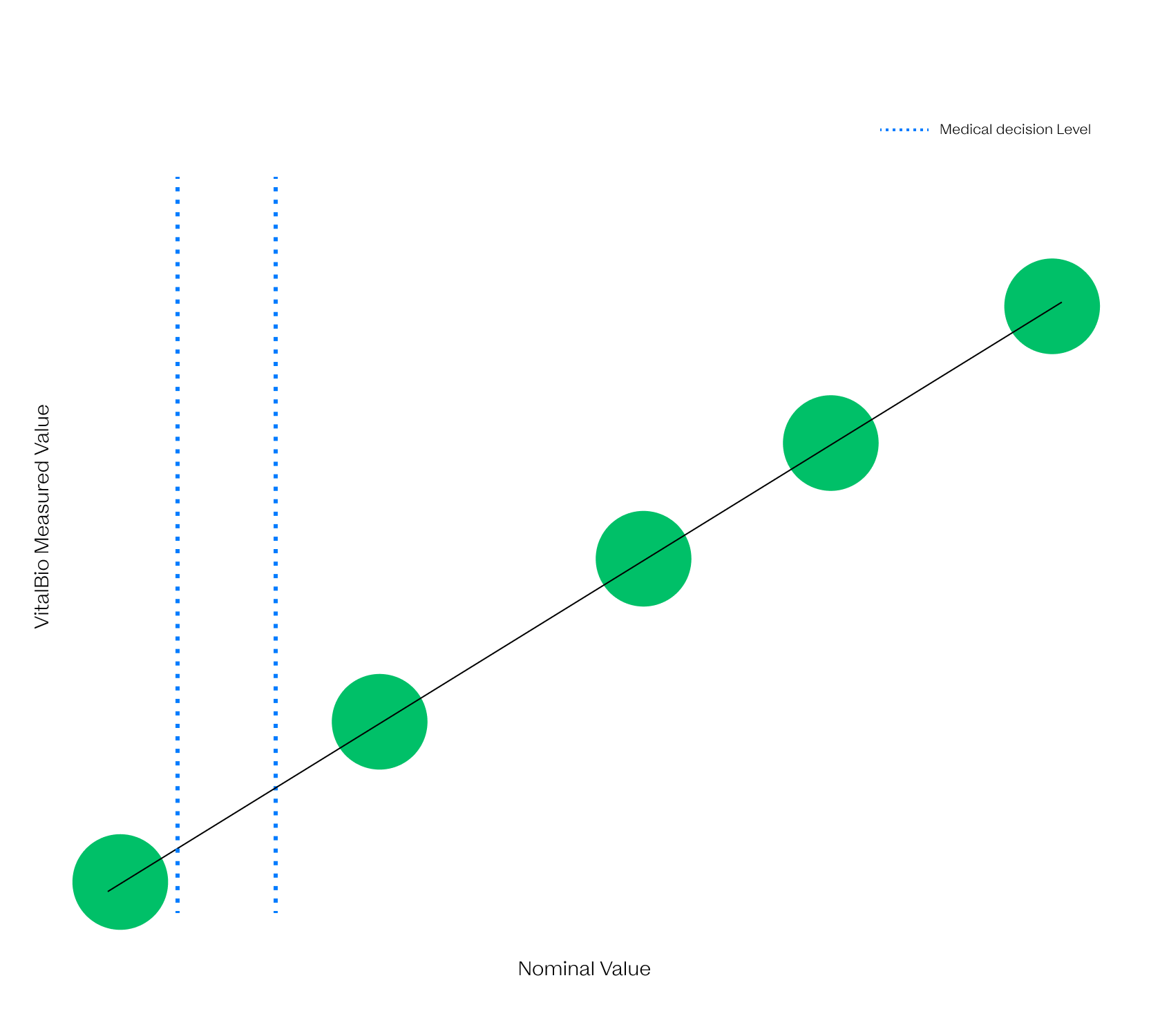
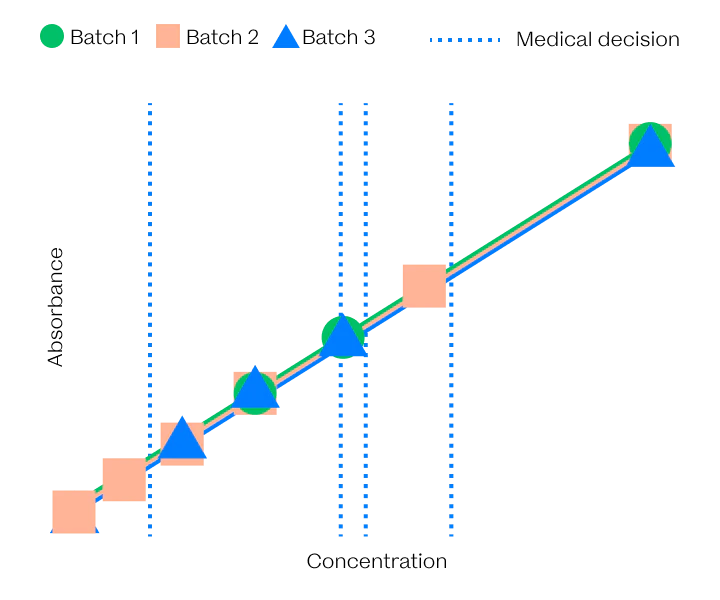
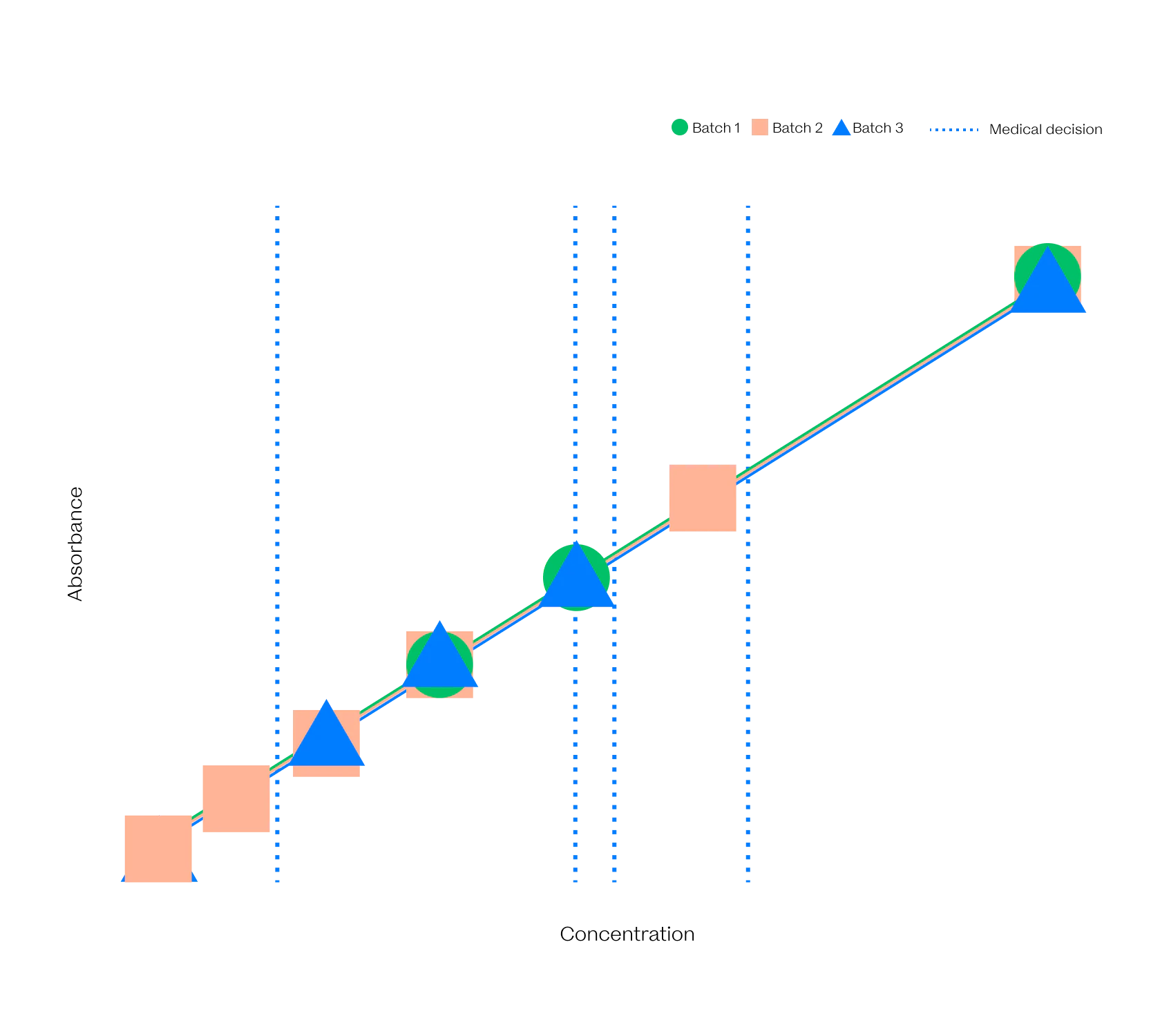
Different in all ways but One
The VitalOne is faster, easier to use, and a lot smaller than its central lab forefathers but intended to give the same high quality results.
As part of our development process, Vital has evaluated production-intent prototype systems against prospectively collected samples and freshly collected samples to put our system to the test against the gold standard.
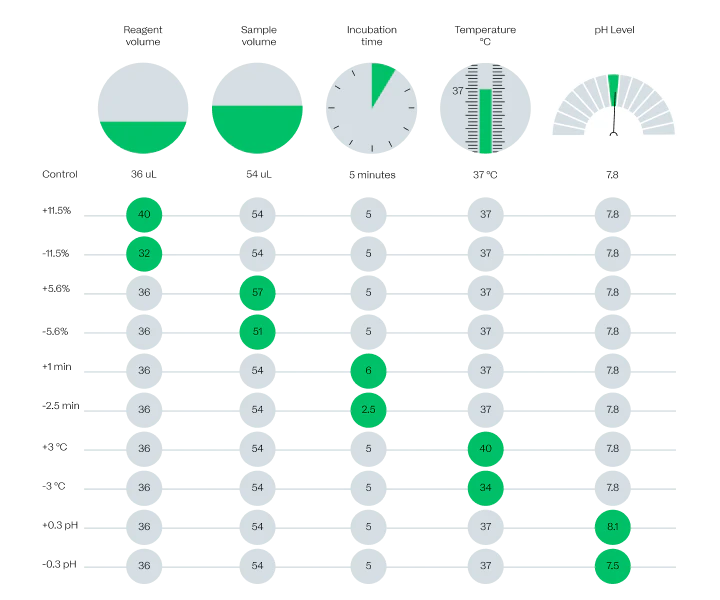
Results you can count on
As part of our development process, we push our assays to the limits to ensure patients will always get the right results.
All Case Studies
Let’s talk healthcare
Vital is transforming diagnostics to ensure patients get the care they need. Provide your information and we’ll be in touch.

